Departments
03 in vitro I (biomaterial)
03 in vitro I: Biomaterial Synthesis & Characterization
Introduction
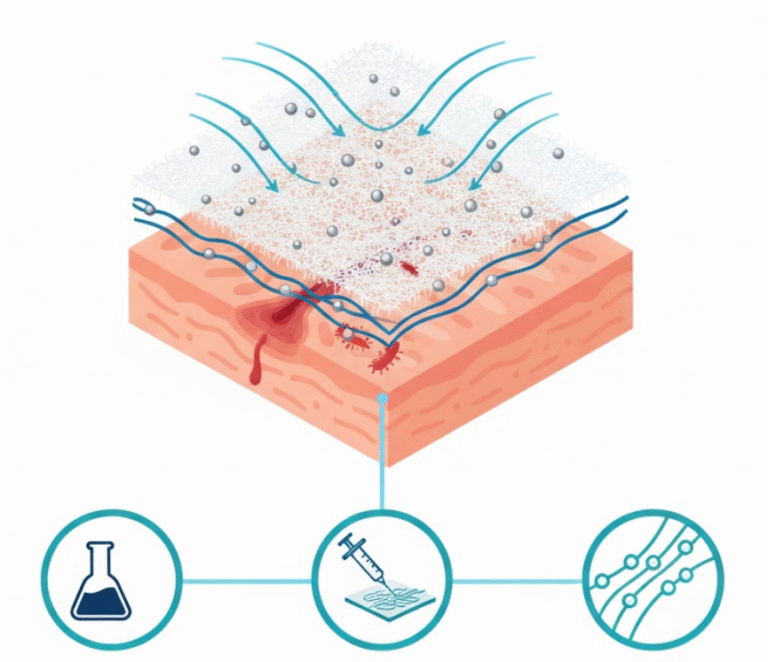
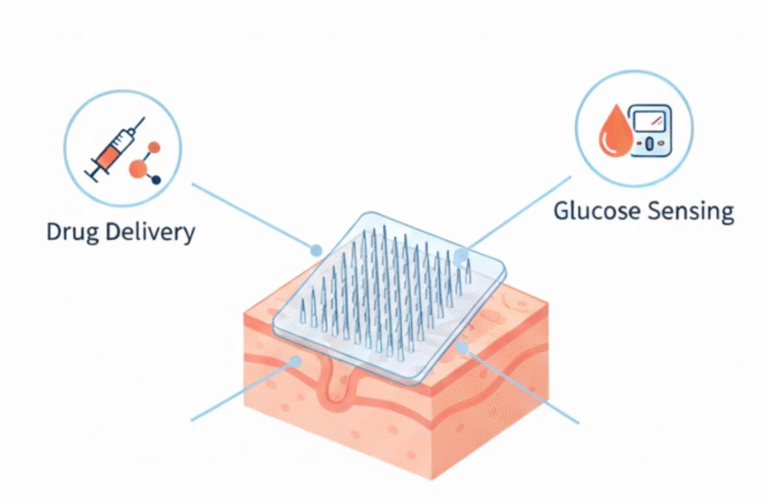
The 03 in vitro I (biomaterial) department at INVITROVO is dedicated to the end-to-end synthesis, development, and characterization of advanced biomaterials. We manage the entire process, from initial formulation through to detailed physicochemical testing, ensuring each step meets the highest standards of quality and precision. Our specialized expertise focuses on creating materials for applications in advanced drug delivery systems and tissue engineering. We handle a wide spectrum of innovative structures, including polymeric scaffolds, hydrogels, drug-loaded nanoparticles, and microneedles. The material’s functional performance and biocompatibility are subsequently evaluated by our in vitro II (cytology) department in comprehensive cellular interaction studies.
Aim
The strategic aim of the 03 in vitro I (biomaterial) department is to optimize material efficacy and performance by providing a reliable foundation for biological validation. We achieve this by focusing on three core objectives:
- To Develop Tailored Materials: To design and fabricate a diverse array of innovative biomaterials, such as polymeric scaffolds and hydrogels, specifically customized for high-impact applications in tissue engineering and drug delivery systems.
- To Validate Material Integrity: To ensure all natural and/or synthesized materials undergo detailed physicochemical characterization before subsequent biological testing, verifying purity, composition, and structural integrity.
- To Enable Translational Success: To ensure materials are optimized and validated under controlled conditions, establishing the crucial performance baseline required before proceeding to in vivo or clinical applications.
Scope
The operational scope of the 03 in vitro I (biomaterial) department encompasses the entire development and analytical pipeline for biomedical materials, from fabrication to comprehensive characterization. We provide full-service support covering:
- Fabrication & Synthesis Strategies:
- Developing specialized structures, including Drug delivery substrates and Tissue engineering scaffolds.
- Preparing Biomaterials and specialized Cell culture medium and substrate.
- Comprehensive Analytical Characterization:
- Physical Testing: Measuring Rheology, Elasticity, Fatigue, Corrosion, and Compression ∣
- Chemical Analysis: Determining Swelling and Contact angle.
- Biomedical Performance: Evaluating Degradation ∣ Biodegradation, Hemolysis, Erosion ∣ Bioerosion, and Drug loading ∣
- Advanced Instrumentation & Methodology:
- Utilizing Spectroscopic Analysis (e.g., FTIR, XRD ∣ XRF ∣ EDX, DLS, TGA) to confirm molecular structure and composition.
- Performing Microscopic Investigations (e.g., SEM, TEM, AFM) for morphological and surface characterization.
- Conducting Chromatography Tests (e.g., HPLC, GPC) to verify purity and component composition.
Meet the Experts

Mohsen Lashgari

Zahra Azizi

Mostafa Bashiri

Prof. Dr.-Ing. Ali Poorkhalil

Sara Rahmati
Offered Services
03 in vitro I (Biomaterial)
• Characterization
• Statistical analysis
•And More