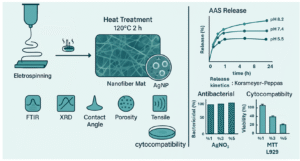
In recent years, the development of smart wound dressings with controlled drug release and antibacterial properties has attracted significant attention, particularly for the treatment of chronic and infected wounds. In this study, a wound dressing based on the Eudragit® L100-55 copolymer combined with chitosan and polycaprolactone (PCL) was fabricated and synthesized. The dressing was prepared using the uniaxial electrospinning method, and during the process, silver nanoparticles (AgNPs) were generated in situ from silver nitrate under thermal treatment within the polymeric matrix. This approach resulted in uniform fibers with suitable morphology and a homogeneous distribution of nanoparticles throughout the polymer network.
In Press. Check back for updates!
